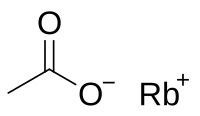
Rubidium acetate
 | |
| Names | |
|---|---|
| IUPAC name Rubidium acetate | |
Other names
| |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.008.415 |
| EC Number |
|
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
| Molar mass | 144.51 g/mol |
| Appearance | White solid |
| Melting point | 246 °C (475 °F; 519 K) (decomposes) |
Solubility in water | 85 g/100 ml (45 °C)[2] |
| log P | -0.561 |
| Hazards | |
| GHS labelling: | |
Hazard statements | H305, H315 |
| NFPA 704 (fire diamond) |  0 1 1 |
| NIOSH (US health exposure limits): | |
PEL (Permissible) | TWA 1 mg/m3 |
| Related compounds | |
Other anions | rubidium formate |
Other cations | Hydrogen acetate Lithium acetate Sodium acetate Potassium acetate Caesium acetate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references | |
Chemical compound
Rubidium acetate is a rubidium salt that is the result of reacting rubidium metal, rubidium carbonate, or rubidium hydroxide with acetic acid. It is soluble in water like other acetates.[2]
Uses
Rubidium acetate is used as a catalyst for the polymerization of silanol terminated siloxane oligomers.[5]
References
- v
- t
- e
 | This catalysis article is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e












