Plutonium(III) chloride 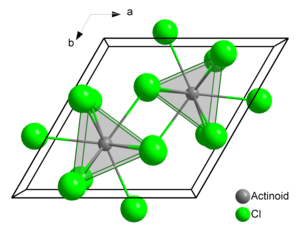 |
| Names |
| IUPAC name Plutonium(III) chloride |
| Other names Plutonium trichloride |
| Identifiers |
CAS Number | - 13569-62-5
 Y[PubChem] Y[PubChem]
|
3D model (JSmol) | |
| ChemSpider | - 14483818
 Y Y
|
| | |
| | - DTXSID90601161

|
InChI=1S/3ClH.Pu/h3*1H;/q;;;+3/p-3  Y YKey: CYMMZQWRMUVJRR-UHFFFAOYSA-K  Y YInChI=1S/3ClH.Pu/h3*1H;/q;;;+3/p-3 Key: CYMMZQWRMUVJRR-DFZHHIFOAR Key: CYMMZQWRMUVJRR-UHFFFAOYSA-K
|
| |
| Properties |
Chemical formula | Cl3Pu |
| Molar mass | 350.322 g/mol |
| Appearance | Green solid |
| Density | 5.71 g/cm3, solid[1] |
| Melting point | 767 °C (1,413 °F; 1,040 K)[1] |
| Boiling point | 1,767 °C (3,213 °F; 2,040 K)[1] |
| Related compounds |
Other anions | PuCl4, PuBr3, SmCl3 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references |
Chemical compound
Plutonium(III) chloride is a chemical compound with the formula PuCl3. This ionic plutonium salt can be prepared by reacting the metal with hydrochloric acid.
Structure
Plutonium atoms in crystalline PuCl3 are 9 coordinate, and the structure is tricapped trigonal prismatic. It crystallizes as the trihydrate, and forms lavender-blue solutions in water.[2]
Safety
As with all plutonium compounds, it is subject to control under the Nuclear Non-Proliferation Treaty. Due to the radioactivity of plutonium, all of its compounds, PuCl3 included, are warm to the touch. Such contact is not recommended, since touching the material may result in serious injury.
References
- ^ a b c www.webelements.com: Plutonium(III) chloride.
- ^ John H. Burns, J. R. Peterson, J. N. Stevenson: "Crystallographic Studies of some Transuranic Trihalides: 239PuCl3, 244CmBr3, 249BkBr3 and 249CfBr3", Journal of Inorganic and Nuclear Chemistry 1975, 37 (3), 743–749; doi:10.1016/0022-1902(75)80532-X.
| Plutonium(II) | |
|---|
| Plutonium(III) | - PuAs
- PuH3
- PuP
- PuB
- PuF3
- PuCl3
- PuBr3
- PuI3
- PuN
|
|---|
| Plutonium(IV) | - PuC
- Pu(NO3)4
- PuF4
- PuO2
- Pu(IO3)4
- Pu(C8H8)2
|
|---|
| Plutonium(V) | |
|---|
| Plutonium(VI) | |
|---|
| Plutonium(VIII) | |
|---|
Salts and covalent derivatives of the chloride ion |
|---|
| HCl | | | | He | | LiCl | BeCl2 | B4Cl4
B12Cl12
BCl3
B2Cl4
+BO3 | C2Cl2
C2Cl4
C2Cl6
CCl4
+C
+CO3 | NCl3
ClN3
+N
+NO3 | ClxOy
Cl2O
Cl2O2
ClO
ClO2
Cl2O4
Cl2O6
Cl2O7
ClO4
+O | ClF
ClF3
ClF5 | Ne | | NaCl | MgCl2 | AlCl
AlCl3 | Si5Cl12
Si2Cl6
SiCl4 | P2Cl4
PCl3
PCl5
+P | S2Cl2
SCl2
SCl4
+SO4 | Cl2 | Ar | | KCl | CaCl
CaCl2 | | ScCl3 | TiCl2
TiCl3
TiCl4 | VCl2
VCl3
VCl4
VCl5 | CrCl2
CrCl3
CrCl4 | MnCl2
MnCl3 | FeCl2
FeCl3 | CoCl2
CoCl3 | NiCl2 | CuCl
CuCl2 | ZnCl2 | GaCl
GaCl3 | GeCl2
GeCl4 | AsCl3
AsCl5
+As | Se2Cl2
SeCl2
SeCl4 | BrCl | Kr | | RbCl | SrCl2 | | YCl3 | ZrCl2
ZrCl3
ZrCl4 | NbCl3
NbCl4
NbCl5 | MoCl2
MoCl3
MoCl4
MoCl5
MoCl6 | TcCl3
TcCl4 | RuCl2
RuCl3
RuCl4 | RhCl3 | PdCl2 | AgCl | CdCl2 | InCl
InCl2
InCl3 | SnCl2
SnCl4 | SbCl3
SbCl5 | Te3Cl2
TeCl2
TeCl4 | ICl
ICl3 | XeCl
XeCl2
XeCl4 | | CsCl | BaCl2 | * | LuCl3 | HfCl4 | TaCl3
TaCl4
TaCl5 | WCl2
WCl3
WCl4
WCl5
WCl6 | ReCl3
ReCl4
ReCl5
ReCl6 | OsCl2
OsCl3
OsCl4
OsCl5 | IrCl2
IrCl3
IrCl4 | PtCl2
PtCl4 | AuCl
(Au[AuCl4])2
AuCl3 | Hg2Cl2
HgCl2 | TlCl
TlCl3 | PbCl2
PbCl4 | BiCl3 | PoCl2
PoCl4 | AtCl | Rn | | FrCl | RaCl2 | ** | LrCl3 | RfCl4 | DbCl5 | SgO2Cl2 | BhO3Cl | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | | | | | * | LaCl3 | CeCl3 | PrCl3 | NdCl2
NdCl3 | PmCl3 | SmCl2
SmCl3 | EuCl2
EuCl3 | GdCl3 | TbCl3 | DyCl2
DyCl3 | HoCl3 | ErCl3 | TmCl2
TmCl3 | YbCl2
YbCl3 | | ** | AcCl3 | ThCl3
ThCl4 | PaCl4
PaCl5 | UCl3
UCl4
UCl5
UCl6 | NpCl3 | PuCl3 | AmCl2
AmCl3 | CmCl3 | BkCl3 | CfCl3
CfCl2 | EsCl2
EsCl3 | FmCl2 | MdCl2 | NoCl2 | |
|
|---|
| | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | | +6 | | | | UF6
UCl6 | NpF6 | PuF6 | AmF6 | | | | EsF6 | | +5 | | | PaF5
PaCl5
PaBr5
PaI5 | UF5
UCl5
UBr5 | NpF5 | PuF5 | | | | | | | +4 | | ThF4
ThCl4
ThBr4
ThI4 | PaF4
PaCl4
PaBr4
PaI4 | UF4
UCl4
UBr4
UI4 | NpF4
NpCl4
NpBr4 | PuF4 | AmF4 | CmF4 | BkF4 | CfF4 | EsF4 | | +3 | AcF3
AcCl3
AcBr3
AcI3 | ThF3
ThCl3
ThI3 | | UF3
UCl3
UBr3
UI3 | NpF3
NpCl3
NpBr3
NpI3 | PuF3
PuCl3
PuBr3
PuI3 | AmF3
AmCl3
AmBr3
AmI3 | CmF3
CmCl3
CmBr3
CmI3 | BkF3
BkCl3
BkBr3
BkI3 | CfF3
CfCl3
CfBr3
CfI3 | EsF3
EsCl3
EsBr3
EsI3 | | +2 | | ThI2
ThCl2 | | | | | AmF2
AmCl2
AmBr2
AmI2 | | | CfI2
CfCl2 | EsCl2
EsBr2
EsI2 | |
 | This inorganic compound–related article is a stub. You can help Wikipedia by expanding it. |

 N verify (what is
N verify (what is  Y
Y N ?)
N ?) 












