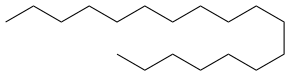
Octadecane
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name Octadecane | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChEBI |
|
| ChemSpider |
|
| ECHA InfoCard | 100.008.902 |
| EC Number |
|
| MeSH | C022883 |
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | C18H38 |
| Molar mass | 254.494 |
| Appearance | White crystals or powder |
| Odor | Odorless |
| Density | 0.777 g mL−1 |
| Melting point | 28 to 30 °C (82 to 86 °F; 301 to 303 K) |
| Boiling point | 317 °C (603 °F; 590 K) |
| Vapor pressure | 1 mm Hg at 119 °C |
Henry's law constant (kH) | 1.9X10-2 atm m3 mol−1 (est) [1] |
Refractive index (nD) | 1.4390 at 20 °C [2] |
| Hazards | |
| Flash point | 165 °C (329 °F; 438 K) |
Autoignition temperature | 235 °C (455 °F; 508 K) |
| Related compounds | |
Related alkanes | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references | |
Chemical compound
Octadecane is an alkane hydrocarbon with the chemical formula CH3(CH2)16CH3.
Properties
Octadecane is distinguished by being the alkane with the lowest carbon number that is unambiguously solid at room temperature and pressure.
References
External links
- Phytochemical and Ethnobotanical Databases
- v
- t
- e
- Methane (CH4)
- Ethane (C2H6)
- Propane (C3H8)
- Butane (C4H10)
- Pentane (C5H12)
- Hexane (C6H14)
- Heptane (C7H16)
- Octane (C8H18)
- Nonane (C9H20)
- Decane (C10H22)
- Undecane (C11H24)
- Dodecane (C12H26)
- Tridecane (C13H28)
- Tetradecane (C14H30)
- Pentadecane (C15H32)
- Hexadecane / Cetane (C16H34)
- Heptadecane (C17H36)
- Octadecane (C18H38)
- Nonadecane (C19H40)
- Icosane (C20H42)
- Heneicosane (C21H44)
- Tetracosane (C24H50)
- Nonacosane (C29H60)
- Hentriacontane (C31H64)
 | This article about a hydrocarbon is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e










