Laurite
H-M symbol: (P 2/m 3)
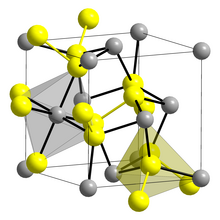
Crystal structure of Laurite
S Ru
Laurite is an opaque black, metallic ruthenium sulfide mineral with formula: RuS2. It crystallizes in the isometric system. It is in the pyrite structural group. Though it's been found in many localities worldwide, it is extremely rare.
Laurite has a Mohs hardness of 7.5 and a specific gravity of 6.43. It can contain osmium, rhodium, iridium, and iron substituting for the ruthenium.[2] The sulfur is present as the disulfide ion, S2−2, so the ruthenium is in the Ru(II) oxidation state.[4]
Discovery and occurrence
It was discovered in 1866 in Borneo, Malaysia and named for Laurie, the wife of Charles A. Joy, an American chemist.[2] It occurs in ultramafic magmatic cumulate deposits and sedimentary placer deposits derived from them. It occurs associated with cooperite, braggite, sperrylite, other minerals of the platinum group elements and chromite.[1]
Synthetic RuS2 is a highly active catalyst for hydrodesulfurization.[5]
References
- ^ a b Anthony, John W.; Bideaux, Richard A.; Bladh, Kenneth W.; Nichols, Monte C. (2005). "Laurite" (PDF). Handbook of Mineralogy. Mineral Data Publishing. Retrieved 14 March 2022.
- ^ a b c Laurite, Mindat.org
- ^ "Laurite Mineral Data". Webmineral.com.
- ^ Cocco, R.A.; Tatarchuk, B.J. (1989). "Effects of presulfidization on the selectivity and surface structure of ruthenium catalysts". Langmuir. 5 (6): 1309–1315. doi:10.1021/la00090a005. Retrieved 20 June 2022.
- ^ Chianelli, R. R.; Berhault, G.; Raybaud, P.; Kasztelan, S.; Hafner, J.; Toulhoat, H. (2002). "Periodic Trends in Hydrodesulfurization: in Support of the Sabatier Principle". Appl. Catal., A. 227 (1–2): 83–96. doi:10.1016/S0926-860X(01)00924-3.
- v
- t
- e
2) and polysulfides
| H2S2 | He | ||||||||||||||||||||
| Li | Be | B | RS2R' | N | S2O | S2F2 | Ne | ||||||||||||||
| Na2Sx | Mg | Al | Si | P | S3 | S2Cl2 | Ar | ||||||||||||||
| K | Ca | Sc | TiS3 | VS4 | Cr | MnS2 | FeS2 | CoS2 | NiS2 | "CuS" | Zn | Ga | Ge | As | Se | S2Br2 | Kr | ||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | RuS2 | Rh | Pd | Ag | Cd | In | Sn | Sb2S5? | Te | S2I2 | Xe | ||||
| Cs | Ba | * | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | |||
| Fr | Ra | ** | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |||
| * | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | |||||||
| ** | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | |||||||
 | This article about a specific sulfide mineral is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e










